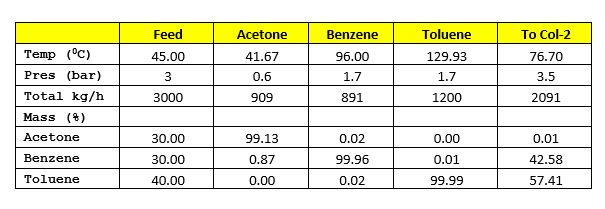
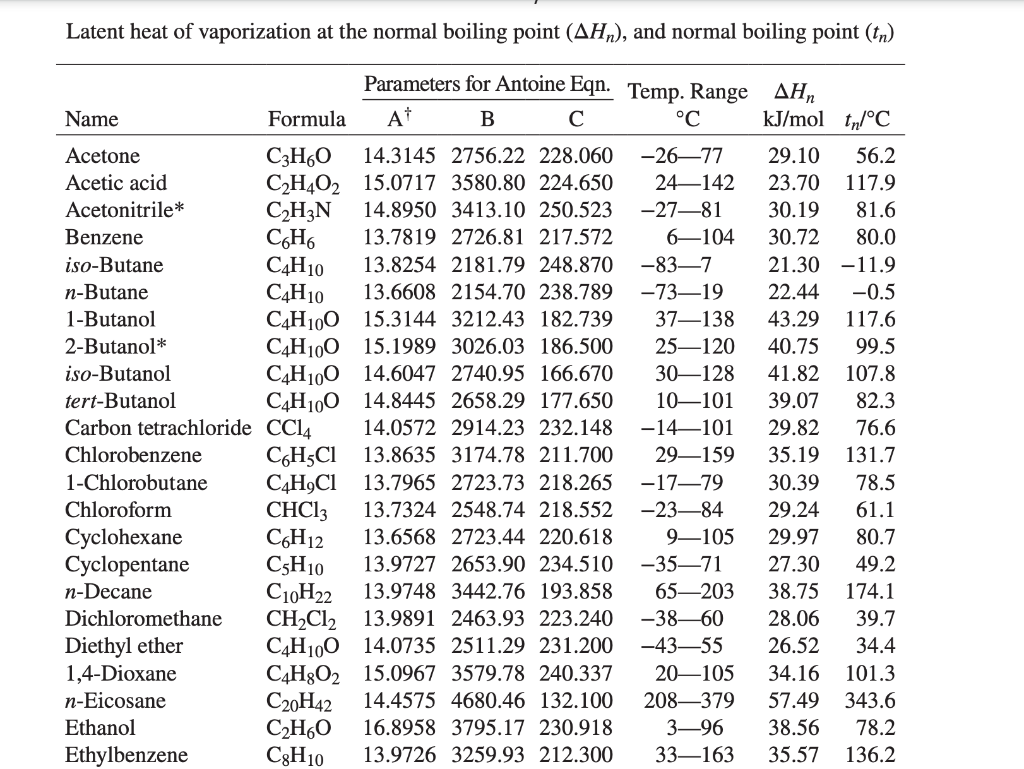
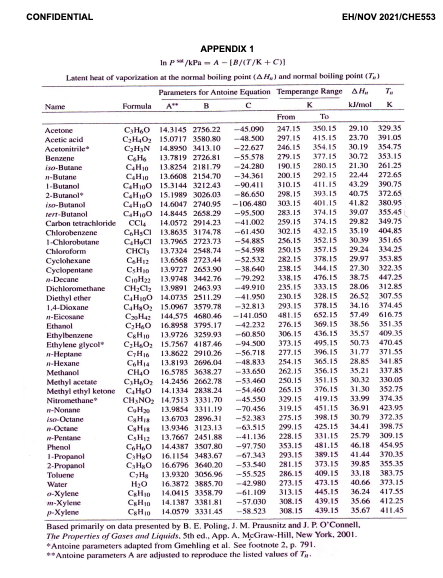
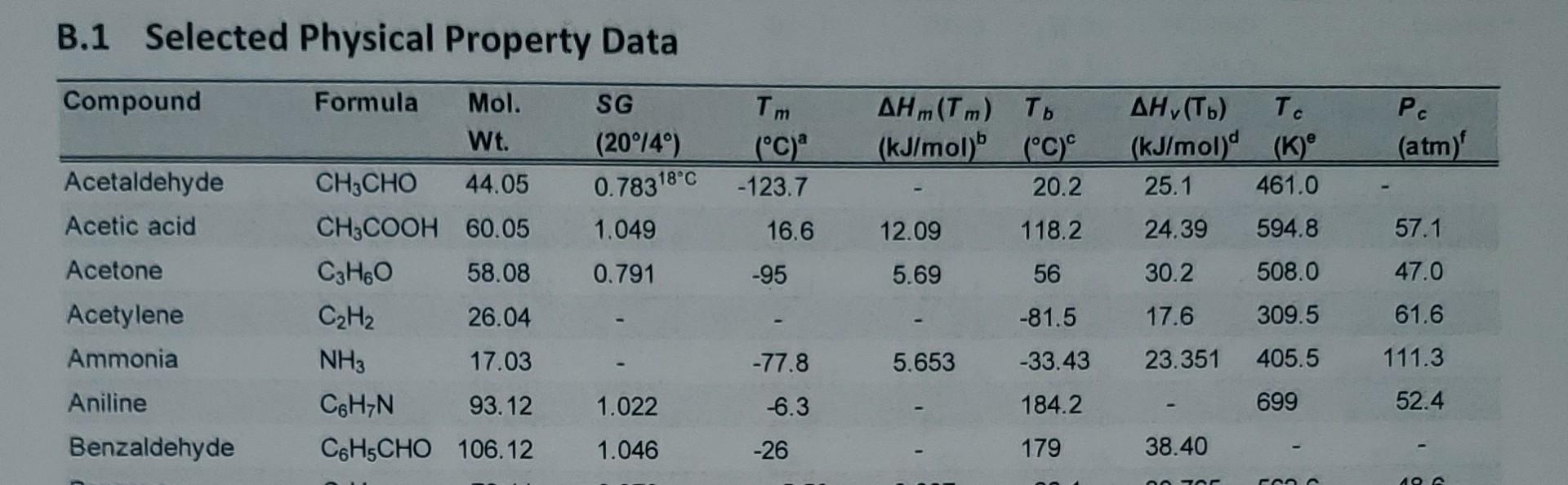
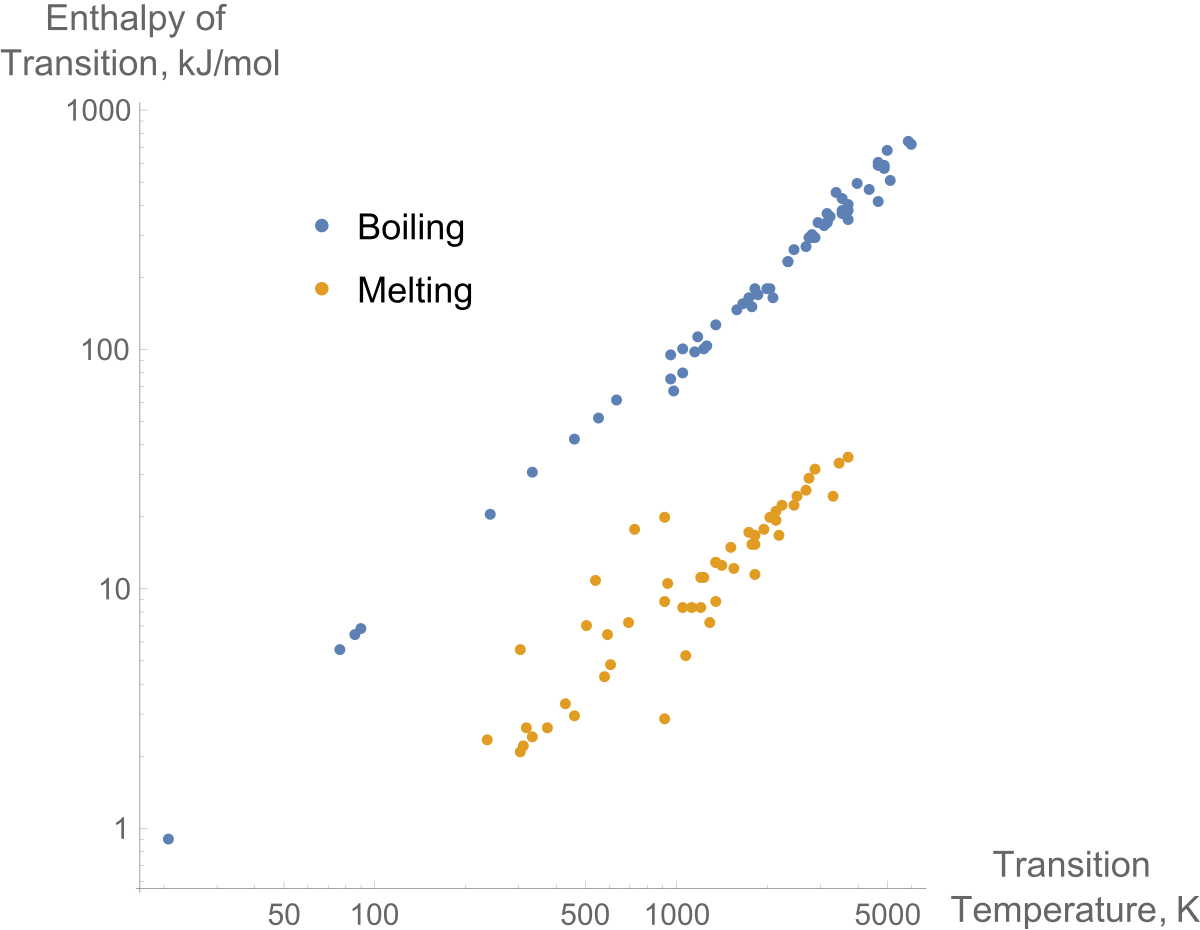
SOLVED: Acetone has an experimentally determined latent heat of vaporization of 30.2 kJ/mol. Use different sources of latent heat and verify with the given experimental value, with the use of Perry's Chemical
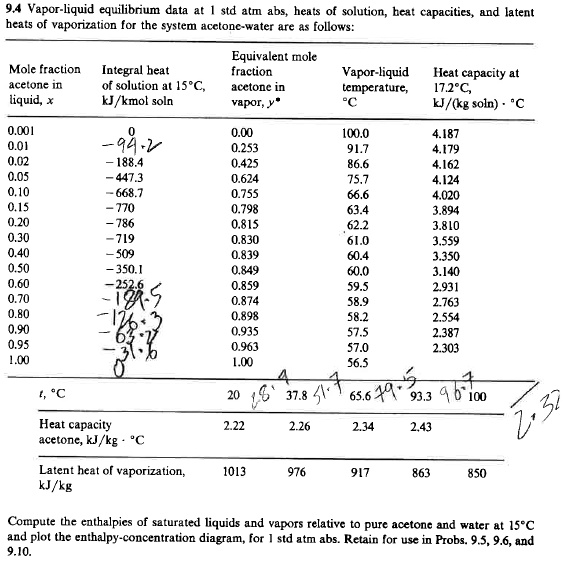
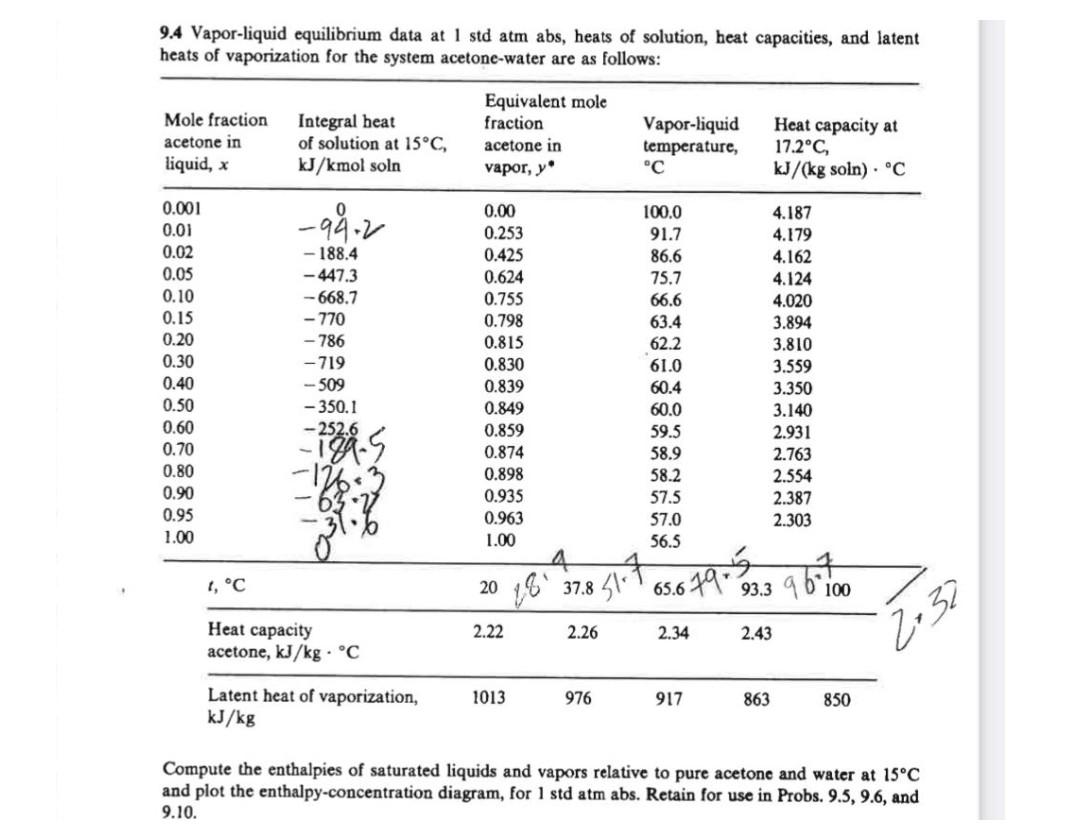
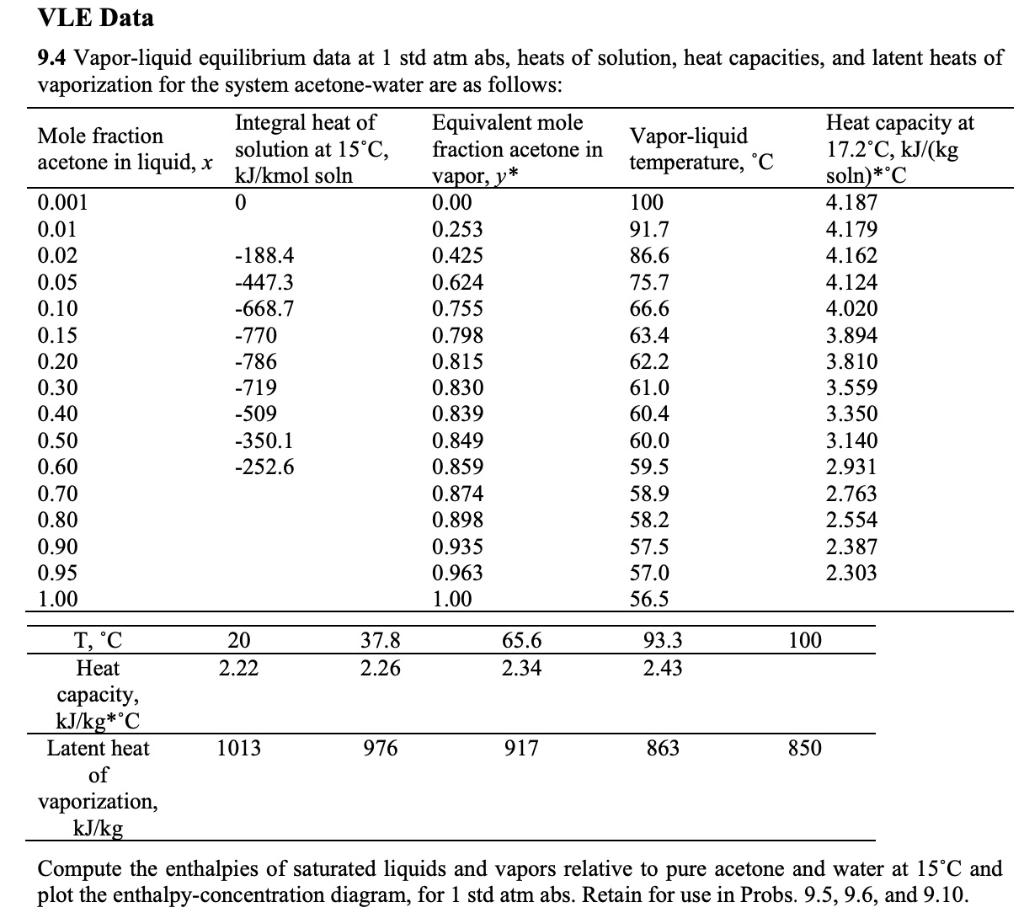
SOLVED: Vapor-liquid equilibrium data at 1 std atm abs, heats of solution, heat capacities, and latent heats of vaporization for the system acetone-water are as follows: compute the enthalpies of saturated liquids
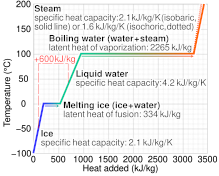
A Theoretical Analysis on Enthalpy of Vaporization: Temperature-Dependence and Singularity at the Critical State Abstract 1. Int

The latent heat of vaporisation of water is 9700 cal/mole and if the b.p. is 100^∘C , the ebullioscopic constant of water is
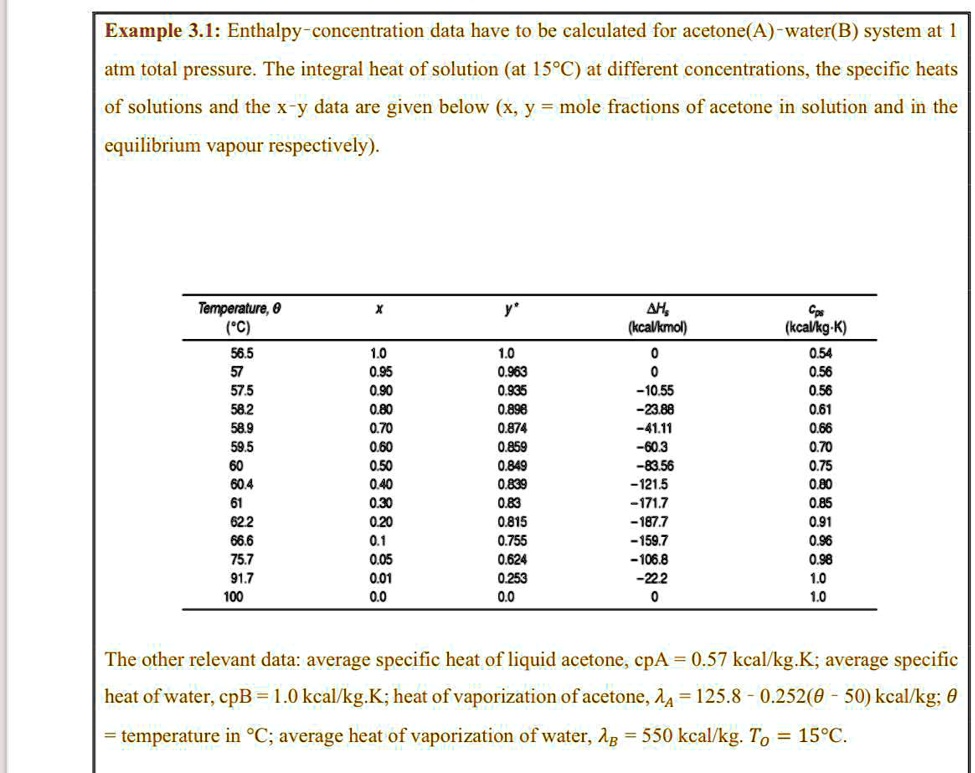
SOLVED: Example 3.1: Enthalpy-concentration data have to be calculated for acetone-water system at 1 atm total pressure. The integral heat of solution at 15°C at different concentrations, the specific heats of solutions,
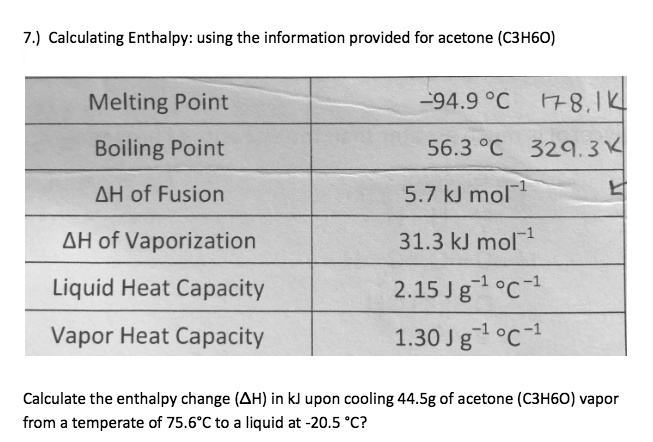
SOLVED: Calculating Enthalpy: using the information provided for acetone (C3H6O) Melting Point Boiling Point AH of Fusion 94.9 °C 56.3 °C 329.34 kJ mol-1 AH of Vaporization 31.3 kJ mol-1 2.15 J

The latent heat of vaporisation of water is 9700 cal/mole and if the b.p. is 100^∘C , the ebullioscopic constant of water is
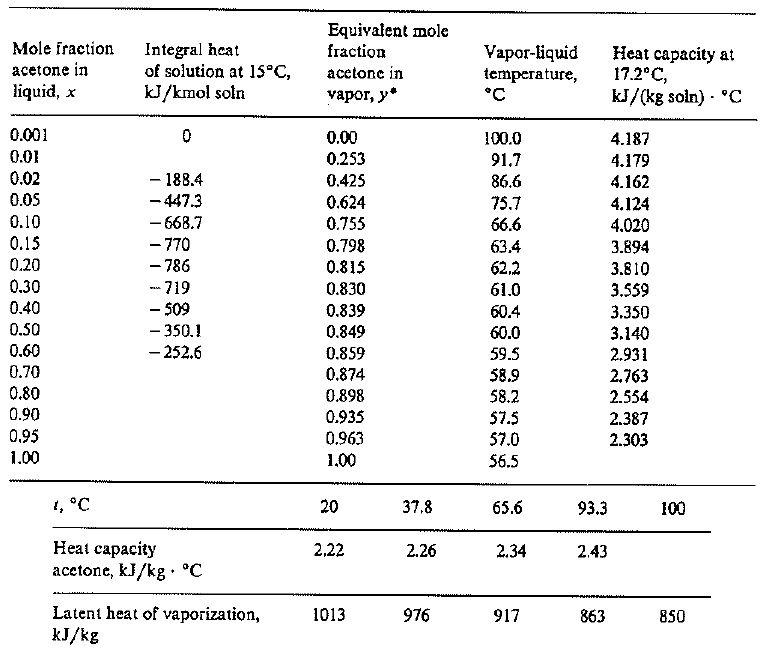
SOLVED: A liquid mixture containing 60 mol% acetone, 40 mol% water, at 26.7°C (80°F), is to be continuously flash-vaporized at 1 std atm pressure, to vaporize 30 mol% of the feed. (a)

Energy efficient design through structural variations of complex heat-integrated azeotropic distillation of acetone-chloroform-water system - ScienceDirect

Variation of effectiveness with mass flow rate of hot fluid (T hi =... | Download Scientific Diagram